Still Exploring?
Looks like you’ve been exploring our platform.
Want to see everything in one place?
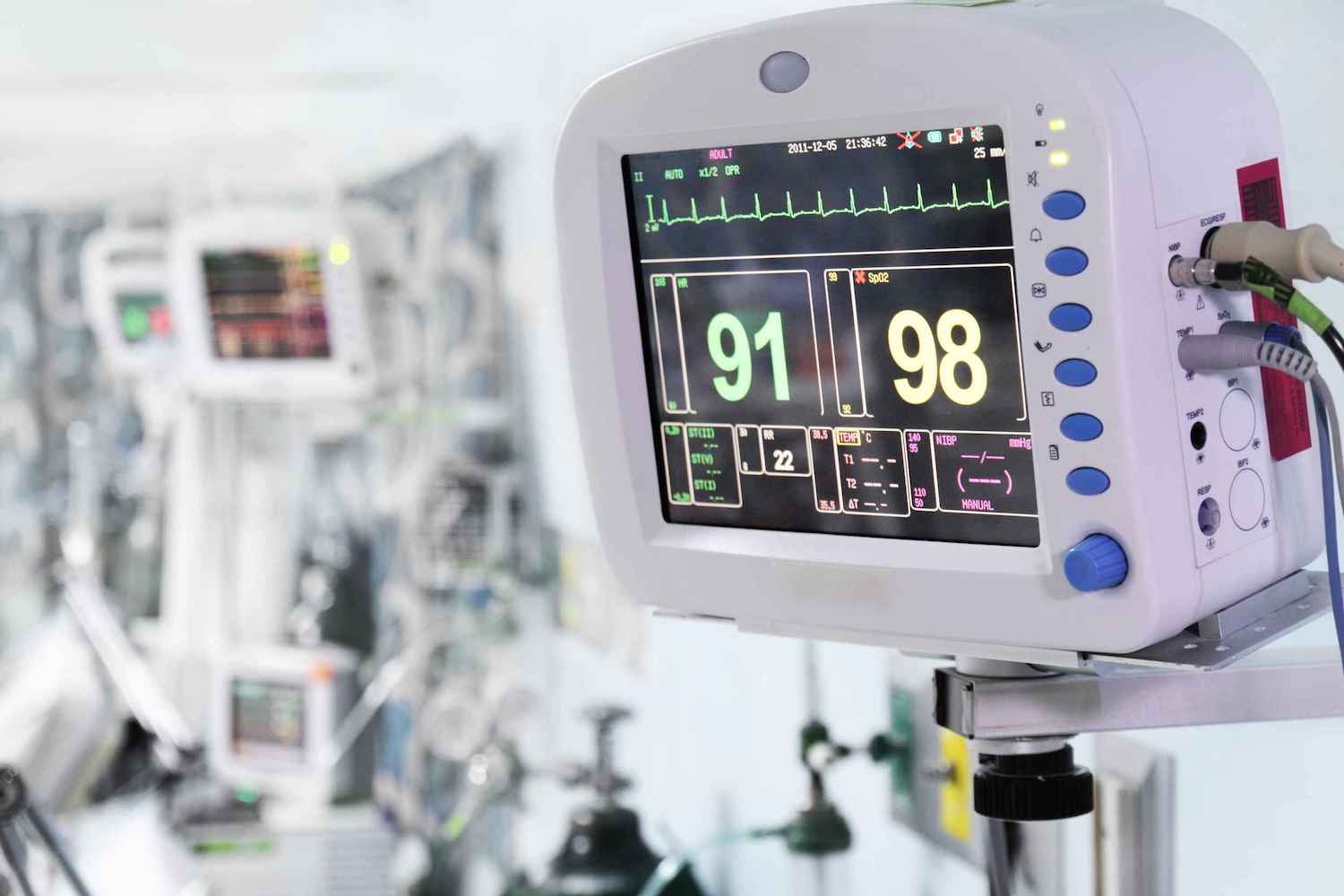
Reduce your risk, protect your EU market access, and increase your program’s productivity under the European Medical Device Regulation (EU MDR) with Assent’s secure MDR software.









Assent’s team of experts ensures your program is up to date with the latest EU MDR requirements, and helps educate your suppliers.
Book a Demo
Automate collection of EU MDR declarations and supply chain data for topics like substances of very high concern (SVHC) and per- and polyfluoroalkyl substances (PFAS).
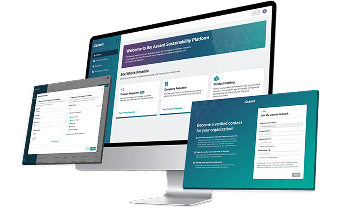
The Assent Sustainability Platform gives medical device manufacturers and their suppliers a centralized and intuitive space to rapidly share compliance data and EU MDR declarations.

Assent makes it easy to roll up medical device supply chain data to generate submissions to the European Database on Medical Devices (EUDAMED).

This FAQ outlines what the EU Medical Device Regulation covers and how Assent’s AI-native software helps manufacturers manage compliance efficiently and accurately.